What is Varithena injectable foam treatment?
LJVascular2024-03-06T13:53:32-08:00Unveiling Varithena: Advancing Vein Health Through Injectable Foam Treatment
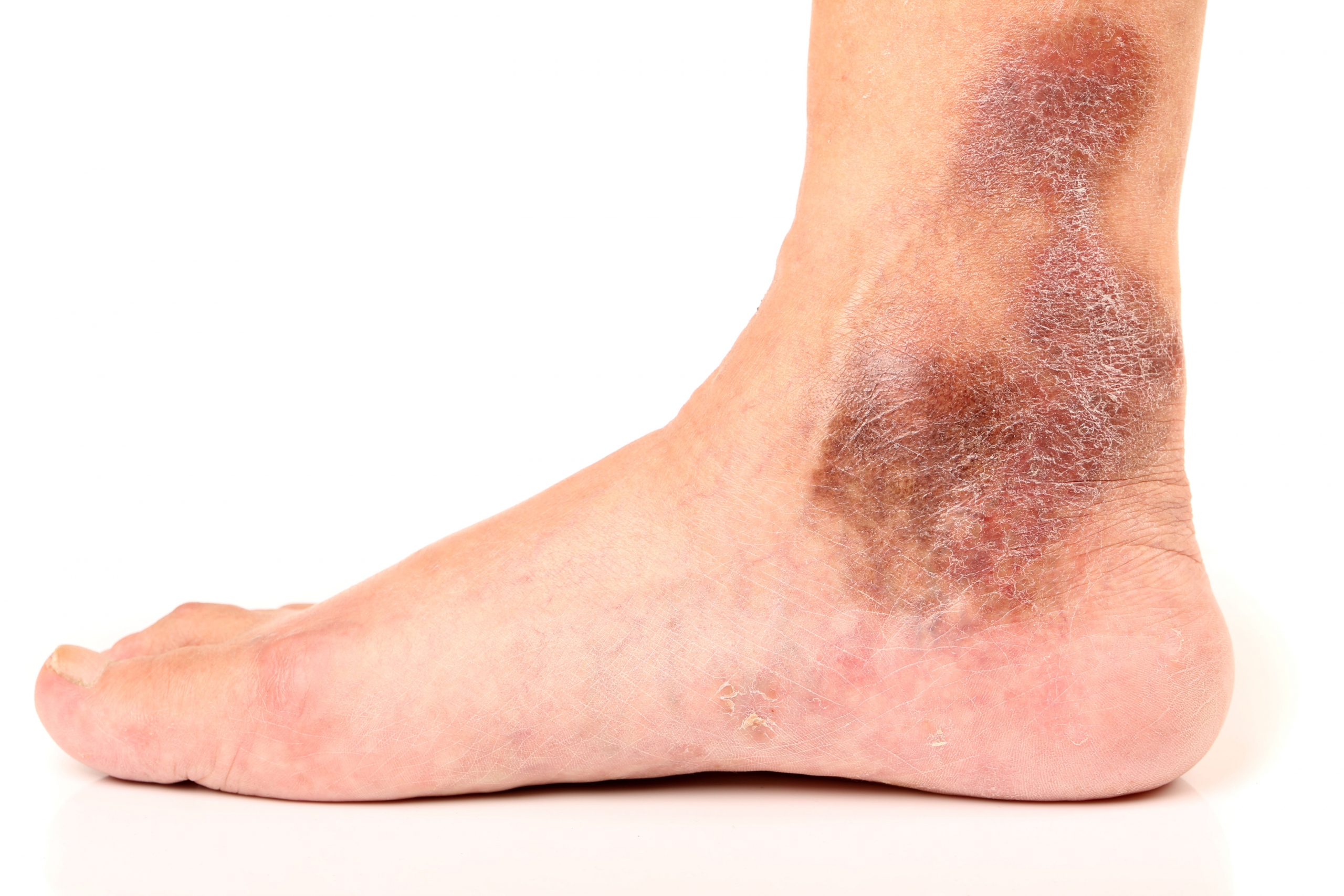
In the landscape of contemporary vein treatments, Varithena (ablation procedure) stands out as a revolutionary solution for managing varicose veins within the great saphenous vein system (GSV). This prescription medication offers a non-thermal, non-tumescent, and nearly painless approach to vein treatment. In this piece, we’ll delve into the intricacies of Varithena®, its efficacy, and what patients can anticipate on the treatment day.
Understanding Varithena®: Your Pathway to Vein Health
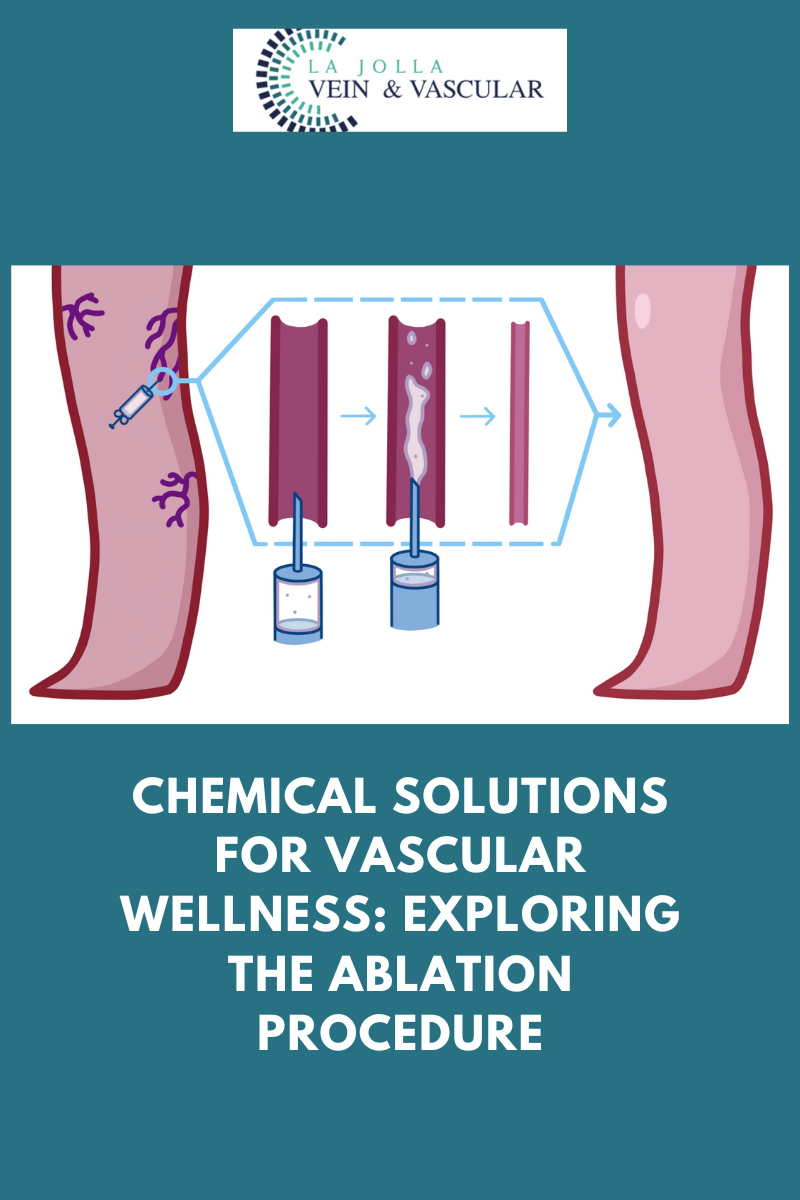
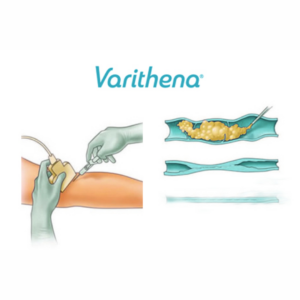
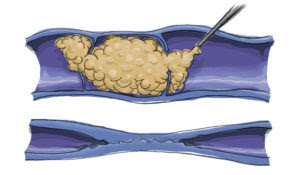
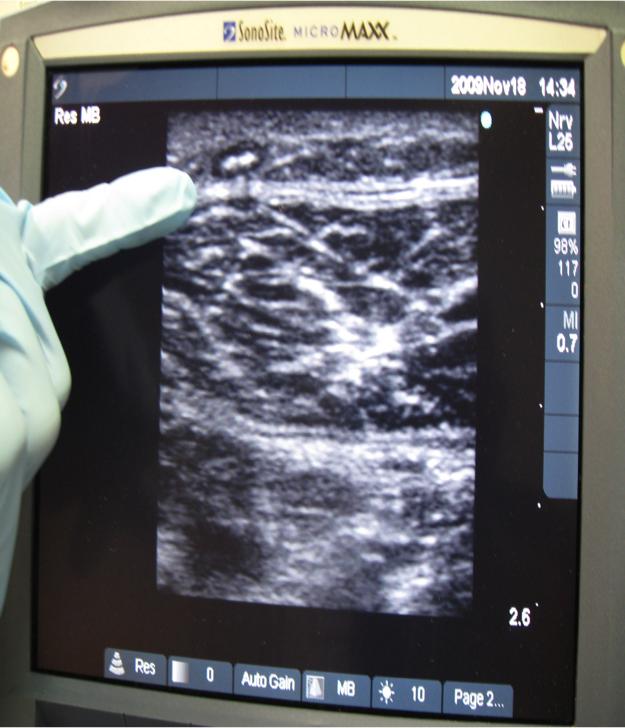
Varithena® employs a “microfoam” formulation comprising polidocanol (sclerosing agent) and CO2 to address superficial venous incompetence and visible varicosities linked to the great saphenous vein system. Skillfully injected into the vein under ultrasound guidance, the microfoam displaces blood and efficiently eliminates the endothelial lining via ablation.
The Benefits of Varithena®:
Tailored Approach: Varithena® recommendations are tailored based on vein location, size, anatomy, and tortuosity, effectively treating veins above and below the knee of varying sizes.
Minimally Invasive: This treatment necessitates no incisions, sedation, or general anesthesia, enhancing patient comfort during the procedure.
Non-Invasive Technique: Unlike other methods, Varithena® doesn’t involve inserting a wire along the length of the vein, reducing associated discomfort.
Heat-Free Process: Varithena® avoids heat application, ensuring no risk of thermal injury during the treatment.
Treatment Frequency and Response: A Detailed Overview
The number and frequency of Varithena® treatments are contingent on individual anatomy, vein response rate, and treatment objectives. While larger or resistant veins might necessitate two treatments for a complete response, many veins typically respond well after a single session. The treatment process is cautiously managed to mitigate potential side effects.
On the Day of Varithena® Treatment: What to Expect
Preparing for your Varithena® ablation treatment is simple:
Consent and Attire: You’ll sign a consent form and change into provided shorts.
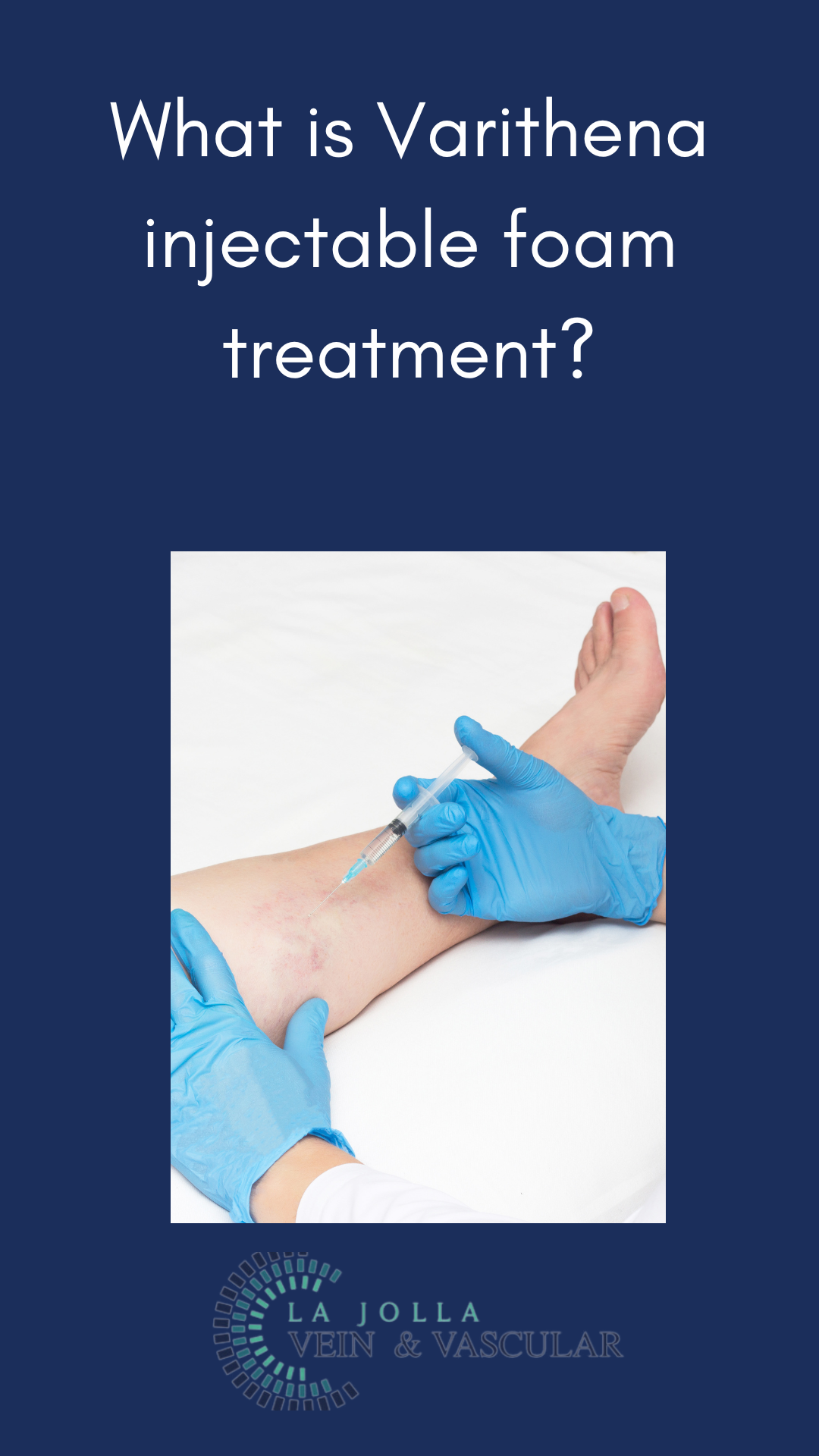
Skin Preparation: Your skin will be cleansed with alcohol, and ultrasound will locate the veins for targeted treatment.
Injection Process: The foam medication will be skillfully injected into your veins using a fine needle. The foam effectively fills and treats the designated vein section, leading to vein collapse and deactivation.
Recovery Period: You’ll elevate your legs on a comfortable wedge pillow for about 15 minutes post-treatment.
Compression Stockings and Walking: Following treatment, compression stockings will be applied, and you’ll walk for 30 minutes before departure.
Varithena® marks a significant stride in vein health, presenting a swift and effective method for managing varicose veins. At La Jolla Vein & Vascular, our proficient medical professionals are committed to providing personalized care throughout your vein health journey. By embracing advanced solutions like Varithena®, you’re embarking on a confident path toward healthier, pain-free legs and improved well-being.
“Bringing Experts Together for Unparalleled Vein and Vascular Care”
La Jolla Vein & Vascular (formerly La Jolla Vein Care) is committed to bringing experts together for unparalleled vein and vascular care.
Nisha Bunke, MD, Sarah Lucas, MD, and Amanda Steinberger, MD are specialists who combine their experience and expertise to offer world-class vascular care.
Our accredited center is also a nationally known teaching site and center of excellence.
For more information on treatments and to book a consultation, please give our office a call at 858-550-0330.
For a deeper dive into vein and vascular care, please check out our Youtube Channel at this link, and our website https://ljvascular.com
For more information on varicose veins and eliminating underlying venous insufficiency,
Please follow our social media Instagram Profile for more fun videos and educational information.
For more blogs and educational content, please check out our clinic’s blog posts!